Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 10, October 2014, Pages 169-173
Original researchOpen Access
Impact of neoadjuvant chemoradiation therapy on the postoperative complication rate in rectal cancer
- 1 Department of Surgery, Charité – Universitätsmedizin Berlin, Campus Benjamin Franklin, Germany
- 2 Department of Gastroenterology, Charité – Universitätsmedizin Berlin, Campus Benjamin Franklin, Germany
- 3 Department of Radiation Oncology and Radiotherapy, Charité – Universitätsmedizin Berlin, Campus Benjamin Franklin, Germany
*Corresponding author: Priv.-Doz. Dr. med. Martin Kruschewski, Klinik für Allgemein- und Viszeralchirurgie, Städtisches Klinikum Solingen gGmbH, Gotenstraße 1, 42653 Solingen, Germany. E-mail: kruschewski.martin@klinikumsolingen.de
Received 10 July 2014 Revised 8 September 2014 Accepted 16 September 2014 Published 23 September 2014
Copyright: © 2014 Kruschewski M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.AbstractTop
Purpose: The impact of neoadjuvant chemoradiation therapy (CRT) on the postoperative complication rate is discussed controversially. Thus the aim of this study was to evaluate the postoperative complication rate in our patient population. Methods: A retrospective analysis was performed to examine all patients documented online who had undergone conventionally fractionated adjuvant or neoadjuvant CRT from 2001 to 2009 in conjunction with curative resection (R0) for sporadic primary colorectal cancer in the middle or lower third. A total of 246 patients were included and analyzed. Two groups were formed: Group I, 2001-2004, adjuvant CRT, n=108, and Group II, 2005-2009, neoadjuvant CRT, n=138. Results: The two groups had comparable patient-, tumor- and therapy-related characteristics. No difference was found in the anastomotic leak rate (Group I vs. II: 10% vs. 11%). The rate of perineal wound healing problems differed significantly (Group I vs. II: 5% vs. 36%, p=0.016). While no patient died in Group I, lethality amounted to 1.5% (2/138) in Group II. Conclusions: Neoadjuvant CRT does not lead to a higher anastomotic leak rate or lethality in comparison to patients who were primarily operated and received adjuvant CRT in the further course. The rate of perineal wound healing problems is significantly increased.
Keywords: rectal cancer; neoadjuvant therapy; adjuvant therapy; chemoradiation therapy; complications; surgery
IntroductionTop
The treatment paradigm for locally advanced and/or node-positive middle and lower third rectal cancer (UICC stage II and III) has shifted from adjuvant to neoadjuvant chemoradiation therapy (CRT) [1, 2]. The impact of this new concept of multimodal rectal cancer therapy on the postoperative complication rate is discussed controversially in the literature [3-15]. Complications are relevant in this connection because they affect not only the immediate postoperative course but also the oncological outcome. Patients without complications after neoadjuvant CRT have a significantly better relapse-free disease and overall survival [16]. Thus the aim of this study was to evaluate the postoperative complication rate in our patient population.
Patients and methodsTop
Study design
A retrospective analysis was performed to examine all patients documented online who had undergone conventionally fractionated adjuvant or neoadjuvant CRT from 2001 to 2009 in conjunction with curative resection (R0) for sporadic primary colorectal cancer in the middle or lower third. Exclusion criteria were multivisceral resections and second primary cancers. A total of 246 patients were included and analyzed. Two groups were formed: Group I, 2001-2004, adjuvant CRT, n=108, and Group II, 2005-2009, neoadjuvant CRT, n=138.
Neoadjuvant or adjuvant chemoradiation
The indication for chemoradiation therapy was the same in both groups. Patients with UICC stage II or III were treated adjuvantly up to 2005 and neoadjuvantly thereafter. The rectal cancer staging required for neoadjuvant therapy was performed by MRI, and endosonography was also used whenever possible.
Treatment was identical in both groups. Radiotherapy consisted of single doses of 1.8 Gy, five times a week, up to a total of 50.4 Gy, delivered (as at least 6-MV photons) in 28 fractions to the pelvis with individual CT-based shaped portals and using of a three-field or four-field box technique. Patients were treated in prone position with a special device (“belly board”) to reduce the dose to the small bowel. The anal sphincter was included in the target volume if the tumor was located in the distal or middle third of the rectum. Fluorouracil (5-FU) was given simultaneously as a continuous infusion with a dose of 225 mg/m2/d throughout the whole treatment, weekends included. The initiation of adjuvant chemoradiation was planned 4 to 6 weeks after the operation (Group I), as the operation was performed 4 to 6 weeks following neoadjuvant chemoradiation (Group II).
Surgical procedure
Both the preoperative preparation and the intervention were standardized. Patients underwent orthograde bowel lavage with polyethylene glycol and received perioperative antibiotic prophylaxis with an aminopenicillin or a cephalosporin plus metronidazole; ampicillin/sulbactam has been used since 2003. Anterior rectal resection was performed according to oncological principles. Total mesorectal excision (TME) was carried out after central ligation of the inferior mesenteric artery and radical lymphadenectomy.
Tumors less than 2 cm from the dentate line were treated by abdominoperineal rectal extirpation with placement of an omental plug to fill the true pelvis. Multilayer closure of the pelvic floor was performed after inserting two 16ch Redon drainage tubes to drain the true pelvis.
Intersphincteric rectal resection was performed for tumors more than 2 cm above the dentate line if the rectum below the tumor could not be closed by a linear stapler. Continuity was restored by colon pouch construction with stapling of colopouch-rectal and suturing of colopouch-anal anastomoses. A protective stoma and pelvic drains were routinely used. The two groups did not differ with regard to intraoperative complications or operating time.
Definition of anastomotic leak
Only clinically manifest and thus relevant anastomotic leaks were recorded. In such cases, fecal matter was discharged from the pelvic drains, surgical wound or vagina or the leaks were detected by computed tomography (CT) with rectal contrast. Indications for CT were: clinical deterioration, abnormal abdominal findings, putrid rectal discharge, turbid drain discharge, and air passage through the drain.
Definition of perineal wound healing problems
Here the term “perineal wound healing problems” subsumes the following manifestations: perineal wound infection, sacral abscess, delayed wound healing or necrosis with subsequent dehiscence (Figure 1).
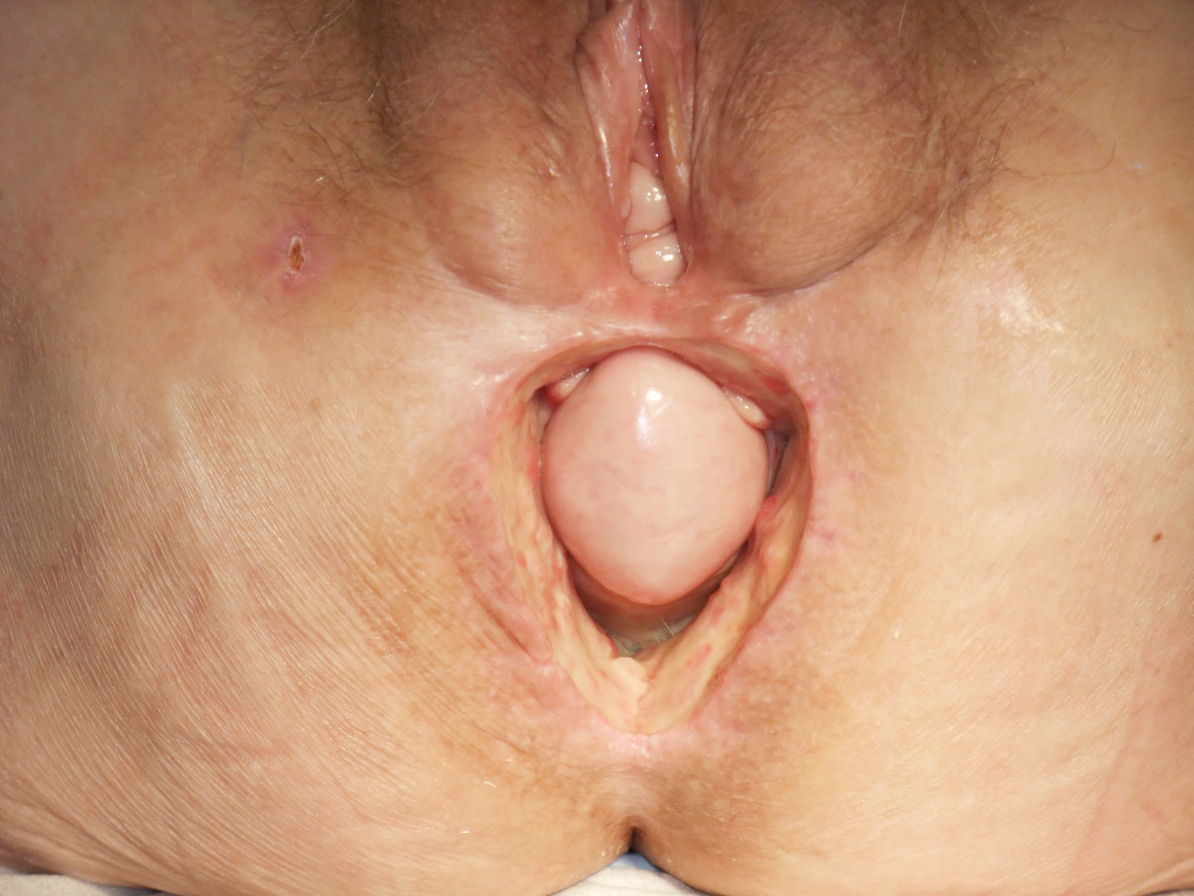
Figure 1 Uterine prolapse in 74-year-old patient with extensive posterior vaginal wall defect s/p perineal wound healing problems s/p rectal extirpation s/p neoadjuvant CRT for T3 lower third rectal cancer.
Statistical analysis
The SPSS computer program was used for statistical analysis. Data were analyzed with the Wilcoxon test, Fisher’s exact test and the chi-square test. Differences with <5% error probability (p<0.05) were considered statistically significant.
Power calculations for Fisher’s exact test were done by drawing 1000 random samples from the binomial distribution. The calculations were implemented and executed using the statistical language R.
ResultsTop
The two groups had comparable patient-, tumor- and therapy-related characteristics. Rectal resections were performed in over 80%. Rectal extirpation was necessary in 19% of patients in Group I (adjuvant CRT) and 16% of those in Group II (neoadjuvant CRT) (Table 1).
| Parameters | Group I (n=108) adjuvant CRT | Group II (n=138) neoadjuvant CRT | p |
| Median age (range) | 63 (38-75) | 64 (43-77) | n.s. |
| Gender (n) | |||
| Male | 61 (57%) | 76 (55%) | n.s. |
| Female | 47 (43%) | 62 (45%) | |
| Comorbidity | |||
| ASA | 2.6 ± 0.4 | 2.5 ± 0.3 | n.s. |
| UICC stage (n) | |||
| UICC stage I | 7 (6%) | 11 (8%) | n.s. |
| UICC stage II | 43 (40%) | 52 (38%) | |
| UICC stage III | 58 (54%) | 75 (54%) | |
| Tumor localization (n) | |||
| Middle third | 67 (62%) | 77 (56%) | n.s. |
| Lower third | 41 (38%) | 61 (44%) | |
| Surgical procedure (n) | |||
| Rectal resection | 87 (81%) | 116 (84%) | n.s. |
| Rectal extirpation | 21 (19%) | 22 (16%) | |
The rate of anastomotic leaks did not differ (Group I vs. II: 10% vs. 11%). The two groups also had a comparable rate of postoperative bleeding, wound dehiscence (burst abdomen) and abdominal wall abscesses. However, a significant difference was found in the rate of perineal wound healing problems after rectal extirpation (Group I vs. II: 5% vs. 36%, p=0.016) (Table 2).
Nonsurgical complications did not differ between the two groups (Table 2). While no patient died in Group I, lethality amounted to 1.5% (2/138) in Group II. One patient died as a result of an anastomotic leak, and the other one succumbed to aspiration pneumonia (Table 2).
| Complications | Group I (n=108) adjuvant CRT | Group II (n=138) neoadjuvant CRT | p | ||
| n | Exitus letalis (n) | n | Exitus letalis (n) | ||
| Surgical complications | |||||
| Anastomotic leak | 9 10%* |
0 | 13 11%* |
1 1% |
n.s |
| Perineal wound healing problems | 1 5%* |
0 | 8 36%* |
0 | 0.016 |
| Postoperative bleeding | 3 3% |
0 | 3 |
0 | n.s. |
| Burst abdomen | 3 3% |
0 | 4 3% |
0 | n.s. |
| Abdominal wall abscess | 9 8% |
0 | 10 7% |
0 | n.s. |
| Nonsurgical complications | |||||
| Myocardial infarction / Heart failure | 9 8% |
0 | 10 7% |
0 | n.s. |
| Pneumonia | 10 9% |
0 | 12 9% |
1 | n.s. |
| Renal failure | 9 8% |
0 | 12 9% |
0 | n.s. |
*related to resections (n=87 and 116) and extirpations (n=21 and 22)
DiscussionTop
Neoadjuvant CRT has gained acceptance in the treatment of middle and lower third rectal cancer (UICC stage II and III), and various studies have examined its impact on the postoperative complication rate. Some of them deal only with complications after rectal resection [3-5, 10-13], others with those after rectal extirpation [6-9, 14] (Table 3).
It is remarkable that all but two [4, 12] of the studies compare the complication rate of a neoadjuvant CRT group with that of a control group submitted to surgery alone without any other form of multimodal therapy. However, the alternative to neoadjuvant CRT would be adjuvant therapy in a multimodal setting. Such a control group is used only in the two studies [4, 12] and in the present investigation (Table 3).
| Reference | Year | n | Type of radiation | Anastomotic leak | rate in % | Perineal wound healing problem | rate in % | ||
| neoadj. | contr. | neoadj. | contr. | ||||||
| Sauer [4] | 2003 | 805 | long-course | = | 12 | 12* | n.d. | -- | -- |
| Lee [5] | 2008 | 1278 | n.d. | + | 11 | 3.2 | n.d. | -- | -- |
| Martel [3] | 2008 | 54 | long-course | = | 5.6 | 6.6 | n.d. | -- | -- |
| Kim [12] | 2010 | 703 | long-course | = | 3.9 | 3.1* | n.d. | -- | -- |
| Garlipp [11] | 2010 | 2085 | long-course | = | 12.4 | 12.7 | n.d. | -- | -- |
| Denost [10] | 2012 | 422 | long-course | = | 12.7 | 10.8 | n.d. | -- | -- |
| Schiffmann [13] | 2013 | 212 | long-course | + | 26.6 | 9.7 | + | 42.2 | 18.8 |
| Bullard [7] | 2005 | 160 | n.d. | n.d. | -- | -- | + | 47 | 23 |
| Chadwick [6] | 2006 | 94 | short-course | n.d. | -- | -- | + | 41 | 5 |
| Artioukh [8] | 2007 | 38 | long-course | n.d. | -- | -- | + | 39 | 7 |
| El-Gazzaz [9] | 2009 | 696 | n.d. | n.d. | -- | -- | + | 20.5 | 13.5 |
| Zorcolo [14] | 2011 | 157 | short-course | n.d. | -- | -- | = | 14.7 | 14.9 |
| Our results | 2011 | 246 (203 and 43) | long-course | = | 11 | 10* | + | 36 | 5* |
Abbreviations: n.d.= no data; neoadj. = neoadjuvant CRT; contr. = control group (no CRT except *(adjuvant CRT as control group)); + = significantly increased; = = no difference.
The type of neoadjuvant CRT is another aspect to be considered when interpreting the data. Short-course radiation therapy, for example, is associated with a markedly increased postoperative complication rate if surgery is performed outside the noncritical interval of 3 to 10 days thereafter [17]. However, the rate of anastomotic leaks and perineal wound healing problems also appears to be significantly increased after long-course radiation therapy if there is a short interval between neoadjuvant CRT and surgery. Kerr et al. conclude that delaying surgery beyond 8 weeks after neoadjuvant CRT reduces postoperative morbidity without compromising the prognosis [18].
It is noteworthy that three of the twelve studies cited in Table 3 fail to provide substantial data on the type of neoadjuvant CRT. Short-course radiation was used in two and conventional fractionated radiation in seven of the nine other studies. Thus they can only be compared to a limited extent.
The rate of anastomotic leaks in the present study was 11% in the neoadjuvant and 10% in the adjuvant group. Other groups also reported similar results [4, 10, 11]. Only two of the seven studies found a significant difference from the neoadjuvant group in terms of the anastomotic leak rate [5, 13] (Table 3).
Various studies have identified neoadjuvant CRT as an independent risk factor for anastomotic leak [5, 19, 20]. However, there are also numerous studies demonstrating that neoadjuvant CRT does not increase the risk of anastomotic leak [21-25]. The definition of “perineal wound healing problems” was based on the literature in which “perineal wound infections” or “perineal wound complications” are discussed [6-9, 14]. It is noteworthy that, despite all methodological reservations regarding the control group and the type of radiation therapy, the rate of perineal wound healing problems after neoadjuvant CRT is significantly increased in all but one study. It ranges from 14.7 to 47% in the neoadjuvant group as opposed to 5-23% in the respective control group (Table 3).
Apparently neoadjuvant CRT additionally compromises the already problematic wound healing after rectal extirpation, since it leads to cell death and progressive occlusive vasculitis not only in the tumor but also in the tissue surrounding it [7].
ConclusionTop
Compared to patients who were primarily operated and received adjuvant CRT, neoadjuvant CRT: i) does not increase the anastomotic leak rate; ii) increases the rate of perineal wound healing problems; iii) does not increase postoperative lethality.
Acknowledgements
We thank Dr. rer. nat. Jan Budczies, Institute of Pathology, Charité - Campus Mitte, for his support in working up the statistical data.
Conflict of interest
The authors declare no conflict of interest.
ReferencesTop
[1] Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, et al. (2004) Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 351:1731-1740. Article Pubmed
[2] Fietkau R (2010) Rectal cancer: quo vadis, neoadjuvant and adjuvant (chemo) radiotherapy? Int J Colorectal Dis 25:285-287. Article Pubmed
[3] Martel G, Al-Suhaibani Y, Moloo H, Haggar F, Friedlich M, et al. (2008) Neoadjuvant therapy and anastomotic leak after tumor-specific mesorectal excision for rectal cancer. Dis Colon Rectum 51:1195-1201. Article Pubmed
[4] Sauer R, Fietkau R, Wittekind C, Rodel C, Martus P, et al. (2003) Adjuvant vs. neoadjuvant radiochemotherapy for locally advanced rectal cancer: the German trial CAO/ARO/AIO-94. Colorectal Dis 5:406-415. Article Pubmed
[5] Lee WS, Yun SH, Roh YN, Yun HR, Lee WY, et al. (2008) Risk factors and clinical outcome for anastomotic leakage after total mesorectal excision for rectal cancer. World J Surg 32:1124-1129. Article Pubmed
[6] Chadwick MA, Vieten D, Pettitt E, Dixon AR, Roe AM (2006) Short course preoperative radiotherapy is the single most important risk factor for perineal wound complications after abdominoperineal excision of the rectum. Colorectal Dis 8:756-761. Article Pubmed
[7] Bullard KM, Trudel JL, Baxter NN, Rothenberger DA (2005) Primary perineal wound closure after preoperative radiotherapy and abdominoperineal resection has a high incidence of wound failure. Dis Colon Rectum 48:438-443. Article Pubmed
[8] Artioukh DY, Smith RA, Gokul K (2007) Risk factors for impaired healing of the perineal wound after abdominoperineal resection of rectum for carcinoma. Colorectal Dis 9:362-367. Article Pubmed
[9] El-Gazzaz G, Kiran RP, Lavery I (2009) Wound complications in rectal cancer patients undergoing primary closure of the perineal wound after abdominoperineal resection. Dis Colon Rectum 52:1962-1966. Article Pubmed
[10] Denost Q, Laurent C, Paumet T, Quintane L, Martenot M, et al. (2012) Laparoscopic surgery for rectal cancer: preoperative radiochemotherapy versus surgery alone. Surg Endosc 26:1878-1883. Article Pubmed
[11] Garlipp B, Ptok H, Schmidt U, Meyer F, Gastinger I, et al. (2010) Neoadjuvant chemoradiotherapy for rectal carcinoma: effects on anastomotic leak rate and postoperative bladder dysfunction after non-emergency sphincter-preserving anterior rectal resection. Results of the Quality Assurance in Rectal Cancer Surgery multicenter observational trial. Langenbecks Arch Surg 395:1031-1038. Article Pubmed
[12] Kim CW, Kim JH, Yu CS, Shin US, Park JS, et al. (2010) Complications after sphincter-saving resection in rectal cancer patients according to whether chemoradiotherapy is performed before or after surgery. Int J Radiat Oncol Biol Phys 78:156-163. Article Pubmed
[13] Schiffmann L, Wedermann N, Gock M, Prall F, Klautke G, et al. (2013) Intensified neoadjuvant radiochemotherapy for rectal cancer enhances surgical complications. BMC surgery 13:43. Article Pubmed
[14] Zorcolo L, Restivo A, Capra F, Fantola G, Marongiu L, et al. (2011) Does long-course radiotherapy influence postoperative perineal morbidity after abdominoperineal resection of the rectum for cancer? Colorectal Dis 13:1407-1412. Article Pubmed
[15] Ulrich A, Weitz J, Slodczyk M, Koch M, Jaeger D, et al. (2009) Neoadjuvant treatment does not influence perioperative outcome in rectal cancer surgery. Int J Radiat Oncol Biol Phys 75:129-136. Article Pubmed
[16] Stelzmueller I, Zitt M, Aigner F, Kafka-Ritsch R, Jager R, et al. (2009) Postoperative morbidity following chemoradiation for locally advanced low rectal cancer. J Gastrointest Surg 13:657-667. Article Pubmed
[17] Hohenberger W, Lahmer G, Fietkau R, Croner RS, Merkel S, et al. (2009) Neoadjuvant radiochemotherapy for rectal cancer. Chirurg 80:294-302. Article Pubmed
[18] Kerr SF, Norton S, Glynne-Jones R (2008) Delaying surgery after neoadjuvant chemoradiotherapy for rectal cancer may reduce postoperative morbidity without compromising prognosis. Br J Surg 95:1534-1540. Article Pubmed
[19] Jestin P, Pahlman L, Gunnarsson U (2008) Risk factors for anastomotic leakage after rectal cancer surgery: a case-control study. Colorectal Dis 10:715-721. Article Pubmed
[20] Matthiessen P, Hallbook O, Andersson M, Rutegard J, Sjodahl R (2004) Risk factors for anastomotic leakage after anterior resection of the rectum. Colorectal Dis 6:462-469. Article Pubmed
[21] Bertelsen CA, Andreasen AH, Jorgensen T, Harling H (2010) Anastomotic leakage after anterior resection for rectal cancer: risk factors. Colorectal Dis 12:37-43. Article Pubmed
[22] Kruschewski M, Rieger H, Pohlen U, Hotz HG, Buhr HJ (2007) Risk factors for clinical anastomotic leakage and postoperative mortality in elective surgery for rectal cancer. Int J Colorectal Dis 22:919-927. Article Pubmed
[23] Konishi T, Watanabe T, Kishimoto J, Nagawa H (2006) Risk factors for anastomotic leakage after surgery for colorectal cancer: results of prospective surveillance. J Am Coll Surg 202:439-444. Article Pubmed
[24] Peeters KC, Tollenaar RA, Marijnen CA, Klein Kranenbarg E, Steup WH, et al. (2005) Risk factors for anastomotic failure after total mesorectal excision of rectal cancer. Br J Surg 92:211-216. Article Pubmed
[25] Rullier E, Laurent C, Garrelon JL, Michel P, Saric J, et al. (1998) Risk factors for anastomotic leakage after resection of rectal cancer. Br J Surg 85:355-358. Article Pubmed



