Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 9, September 2014, Pages 144-152
Original researchOpen Access
Adjuvant radiotherapy for melanoma metastatic to axillary and inguinal nodes: Regional tumor control and late effects
-
Safia K. Ahmed1,,
Lisa A. Kottschade2,
Svetomir N. Markovic2,3,4,
Yolanda I. Garces1 and
Robert L. Foote1,*

- 1 Department of Radiation Oncology, Mayo Clinic, Rochester, Minnesota, USA
- 2 Division of Medical Oncology, Mayo Clinic, Rochester, Minnesota, USA
- 3 Division of Hematology, Mayo Clinic, Rochester, Minnesota, USA
- 4 Division of Clinical Immunology and Immunotherapeutics, Mayo Clinic, Rochester, Minnesota, USA
*Corresponding author: Dr. Robert L. Foote, MD, Department of Radiation Oncology, Mayo Clinic, 200 First St SW, Rochester, MN 55905, USA. E-mail: foote.robert@mayo.edu
Received 7 June 2014 Revised 13 August 2014 Accepted 21 August 2014 Published 28 August 2014
DOI: http://dx.doi.org/10.14312/2052-4994.2014-22
Copyright: © 2014 Ahmed SK, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Background: Adjuvant radiotherapy reduces nodal recurrence in metastatic melanoma. The purpose of this study was to examine the effects of adjuvant radiotherapy on in-field nodal control of melanoma metastatic to axillary and inguinal lymph nodes. Methods: The medical records of patients who received adjuvant radiotherapy from January 1, 2003, through December 31, 2011, at Mayo Clinic were reviewed. Results: In the 20 patients identified, the rates of 2-year overall survival, nodal control, and freedom from distant metastases were 53%, 54%, and 44%, respectively. Intensity-modulated radiotherapy was used in 15 patients (75%) and 3-dimensional conformal radiotherapy in 5 (25%). Disease progression was documented in 13 patients, with a total of 10 distant metastases and 6 in-field nodal recurrences. Treatment complications occurred in 14 patients: lymphedema, 9; lymphedema and fibrosis, 1; lymphedema and wound dehiscence, 1; wound dehiscence, 1; lymphedema and seroma, 1; and lymphedema, seroma, and fibrosis, 1. Conclusion: Patients with axillary and inguinal nodal metastases from melanoma have poor overall survival. Surgery and adjuvant radiotherapy provided a 54% nodal control rate. No plexopathy or grade 3 lymphedema complications were seen; however, 20% developed wound dehiscence.
Keywords: adjuvant radiotherapy; axillary; inguinal; lymph node; melanoma; metastasis
IntroductionTop
Surgery has been the standard treatment for melanoma metastatic to lymph nodes [1-4]; however, recent data suggest adjuvant radiotherapy reduces regional nodal recurrence [5]. Regional lymph node recurrence rates range from 5% to 20% after adjuvant radiotherapy compared with 20% to 50% after surgery alone [1-4, 6-11].
Radiotherapy is often not administered because of a belief that melanoma is radioresistant [12]. Studies have suggested high-dose-per-fraction radiotherapy may render melanoma radioresponsive [13]. However, this has not been confirmed by prospective clinical trials [14]. The long-term sequelae of high-dose-per-fraction radiotherapy, especially intensity-modulated radiotherapy (IMRT), in patients with melanoma metastatic to axillary or inguinal lymph nodes have not been thoroughly characterized [7, 12, 14]. The aim of this study was to examine the effects of adjuvant radiotherapy on in-field nodal control (NC) of melanoma metastatic to axillary and inguinal lymph nodes and to document treatment-related complications.
Materials and methodsTop
After approval from the Mayo Clinic Institutional Review Board, patients who received postoperative radiotherapy for melanoma metastatic to axillary or inguinal lymph nodes were identified through a search of the Department of Radiation Oncology tumor registry at Mayo Clinic, Rochester, Minnesota. We started using IMRT in 2003, and the study period spanned January 1, 2003, through December 31, 2011. All patients whose data were used in this study granted permission for their data to be used for research purposes.
Relapse was defined as any clinical or radiographic evidence of tumor recurrence. In-field NC was defined as non-recurrence of lymph node disease within the radiotherapy treatment volume. Out-of-field marginal recurrence was defined as recurrence of lymph node disease outside but adjacent to the radiotherapy treatment volume. Treatment-related lymphedema was retrospectively characterized according to the Common Terminology Criteria for Adverse Events version 4.0. Grade characterization required inference based on documented pertinent positive and negative physical examination findings.
Overall survival, freedom from relapse, freedom from distant metastasis, freedom from out-of-field marginal recurrence, and in-field NC were estimated using the Kaplan-Meier method and compared between subgroups using the log-rank test in univariate analysis. Because of small sample size, multivariate analysis was not performed.
Data were analyzed using JMP statistical software (version 9; SAS Institute Inc, Cary, North Carolina). Chi-squared tests were used to examine associations between categorical variables. A P value of less than 0.05 was considered significant. The start of radiotherapy was used as time 0.
ResultsTop
Patient characteristics
Twenty-four patients received radiotherapy after axillary or inguinal lymph node surgery for metastatic melanoma. Excluded were 4 patients treated palliatively for gross residual disease. A total of 20 patients (11 with axillary lymph node metastases, 9 with inguinal lymph node metastases) were studied. Patient and tumor characteristics at the time of initial melanoma diagnosis are listed in Table 1.
| Characteristic | Value |
| All patients | 20 (100) |
| Age (years) | |
| Median | 53 |
| Range | 28-72 |
| Sex | |
| Male | 14 (70) |
| Female | 6 (30) |
| Site of primary melanoma | |
| Occult primary | 6 (30) |
| Upper extremity | 4 (20) |
| Lower extremity | 4 (20) |
| Back | 3 (15) |
| Chest | 2 (10) |
| Conjunctiva | 1 (5) |
| Breslow thickness, mm | |
| Median | 1.6 |
| Range | 0.4-17 |
| ≤1.00 | 3 (15) |
| 1.01-2.00 | 2 (10) |
| 2.01-4.00 | 5 (25) |
| >4.00 | 4 (20) |
| Unknown primary | 6 (30) |
| Clark level | |
| I | 0 (0) |
| II | 0 (0) |
| III | 1 (5) |
| IV | 7 (35) |
| V | 2 (10) |
| Not documented | 4 (20) |
| Unknown primary | 6 (30) |
| Ulceration | |
| Yes | 7 (35) |
| No | 7 (35) |
| Unknown primary | 6 (30) |
Abbreviations: a = All values are number (percentage) unless indicated otherwise.
Treatment
Sixteen patients (80%) underwent axillary or inguinal lymph node surgery at the time of the initial diagnosis and surgery for the primary tumor and were found to be lymph node positive. The remaining 4 patients (20%) were observed and subsequently developed lymph node metastases. The time to delayed lymph node metastasis was 9 months, 17 months, 22 months, and 31 months. For the 17 patients in whom the size of the largest involved lymph node was documented, the median was 6.0 cm (range, 0.4-8.8 cm). The median number of lymph nodes involved was 2 (range, 1-19). Table 2 lists the nodal characteristics. Nodal staging according to the American Joint Committee on Cancer staging system (seventh edition) [15] was categorized at the time of surgery that preceded radiotherapy. None of the patients had local recurrence at the time of their delayed nodal recurrence.
| Characteristic | Axillary | Inguinal |
| Total | 11 (55) | 9 (45) |
| Status of node disease | ||
| Primary | 8 (73) | 8 (89) |
| Delayed | 3 (27) | 1 (11) |
| Size of largest node, cm | ||
| < 3 | 1 (9) | 5 (56) |
| ≤ 3 | 8 (73) | 3 (33) |
| Unknown | 2 (18) | 1 (11) |
| No. of positive nodes | ||
| ≤2 | 6 (55) | 6 (67) |
| >2 | 5 (45) | 3 (33) |
| Extracapsular extension | ||
| No | 7 (64) | 5 (56) |
| Yes | 4 (36) | 4 (44) |
| Nodal stagingb | ||
| 1a | 0 (0) | 1 (11) |
| 1b | 4 (36) | 4 (44) |
| 2a | 1 (9) | 0 (0) |
| 2b | 3 (27) | 2 (22) |
| 3 | 3 (27) | 2 (22) |
| RT dose, Gy/fractions | ||
| 35/5 | 9 (82) | 3 (33) |
| 35/5 | 1 (9) | 0 (0) |
| 36/6 | 1 (9) | 1 (11) |
| 48/20 | 0 (0) | 5 (56) |
Abbreviations: a = Values are number (percentage); b = American Joint Committee on Cancer [15].
The surgical procedure performed was lymphadenectomy or lymph node dissection in 17 patients (85%) and local lymph node excision in 3 patients (15%). Immunotherapy agents were prescribed to 14 patients, and cytoxic chemotherapy was prescribed to 2 patients. Table 2 lists the radiotherapy dose administered. The median time interval from surgery to the start of postoperative radiotherapy was 5.1 weeks (range, 3.0-10.5 weeks). The median dose was 30 Gy in 5 fractions over 14 days (ranges, 30-48 Gy; 5-20 fractions; 12-29 days). One patient received 35 Gy in 5 fractions (over 14 days) and 2 patients received 36 Gy in 6 fractions (over 14 days) because of suspected microscopic residual disease based on our review of the operative notes, pathology reports, imaging studies, and clinical notes. Treatment of patients who received 30 Gy in 5 fractions or 48 Gy in 20 fractions was considered truly adjuvant.
IMRT was used in 15 patients (75%), and 3-dimensional conformal radiotherapy was used in 5 patients (25%). Thirteen patients (65%) received radiotherapy immediately after initial lymph node surgery, whereas 7 patients (35%) received radiotherapy after surgery for lymph node recurrence or progression after initial lymph node surgery or after initial systemic therapy. Four of 7 patients received radiotherapy on the first nodal recurrence or progression after lymph node surgery or systemic therapy, whereas 1 patient received radiotherapy on the second documented lymph node recurrence after initial lymph node surgery, and 1 patient received radiotherapy on the third documented lymph node recurrence. One patient received systemic therapy for the initial lymph node metastasis and then received surgery and radiotherapy on lymph node progression. Patient nodal characteristics, treatment course, outcomes, and complications are summarized in Table 3.
| Largest LN, cm | ECE | AJCC nodal staging | Time to postoperative RT, wk | RT type | RT dose, Gy/fx | Days of RT | Surgery and postoperative RT timing | Recurrence | Survival | Complications |
| 3 | No | 3b | 5.0 | 3-D | 30/5 | 14 | First line | D | Dead | None |
| 8.3 | No | 1b | 3.3 | 3-D | 30/5 | 12 | First line | D | Dead | L |
| 7.2 | Yes | 3b | 3.0 | IMRT | 30/5 | 14 | First line | N+D | Dead | None |
| 0.4 | Yes | 1b | 5.1 | 3-D | 30/5 | 14 | First line | D | Dead | None |
| 8 | Yes | 2b | 5.6 | IMRT | 30/5 | 14 | First line | D | Dead | WD |
| No information | Yes | 3b | 10.4 | IMRT | 36/6 | 14 | First line | D | Dead | L |
| 6.2 | No | 1b | 6.7 | IMRT | 30/5 | 15 | First line | None | Alive | None |
| 6 | No | 2b | 6.4 | IMRT | 35/5 | 14 | First line | None | Alive | None |
| No information | No | 2a | 6.1 | IMRT | 30/5 | 14 | First line | N | Alive | None |
| 6.5 | No | 2b | 4.6 | IMRT | 30/5 | 14 | First line | None | Alive | L |
| 6 | No | 1b | 5.0 | IMRT | 30/5 | 14 | Recurrence | None | Alive | L |
| 5 | No | 1a | 3.7 | IMRT | 48/20 | 24 | First line | None | Alive | L |
| 7.4 | No | 1b | 10.5 | 3-D | 48/20 | 29 | Recurrence | M+D | Dead | L |
| 8.8 | No | 2b | 4.8 | IMRT | 48/20 | 24 | Recurrence | D | Alive | L, fibrosis |
| 2 | Yes | 1b | 6.0 | IMRT | 30/5 | 14 | Recurrence | None | Alive | L |
| 3.5 | Yes | 2b | 5.3 | IMRT | 30/5 | 14 | First line | D | Dead | L |
| 2.8 | No | 3b | 4.8 | IMRT | 48/20 | 25 | Recurrence | M | Dead | L |
| 0.9 | Yes | 3b | 4.0 | IMRT | 48/20 | 25 | Recurrence | None | Alive | L, seroma |
| 2.7 | Yes | 1b | 5.0 | IMRT | 30/5 | 14 | First line | D | Alive | L, WD |
| No information | No | 1b | 6.7 | 3-D | 36/6 | 14 | Recurrence | N | Dead | L, seroma, fibrosis |
Abbreviations: A = axillary lymph node; AJCC = American Joint Committee on Cancer seventh edition; D = distant failure; ECE = extracapsular extension; fx = fractions; I = inguinal; IMRT = intensity-modulated radiotherapy; L = lymphedema; LN = lymph node; M = marginal failure; M+D = marginal and distant failure; N = nodal failure; N+D = nodal and distant failure; P = primary diagnosis; R = diagnosis at time of recurrence; RT = radiotherapy; 3-D = 3-dimensional conformal radiotherapy; WD = wound dehiscence.
Survival outcomes
At the time of analysis, 10 patients were alive, with a median follow-up time of 15 months (range, 6-33 months). The 2-year overall survival, freedom from any relapse, and in-field NC rates were 53% (95% confidence interval [CI], 32%-87%), 27% (95% CI, 12%-60%), and 54% (95% CI, 31%-95%), respectively (Figure 1). The 2-year freedom from distant metastases was 44% (95% CI, 25%-77%) and freedom from out-of-field marginal recurrence was 89% (95% CI, 77%-100%) (Figure 1). The 2-year NC rate was 54% and 67% (at 19 months) for patients with axillary metastases and inguinal metastases, respectively (P = .99).
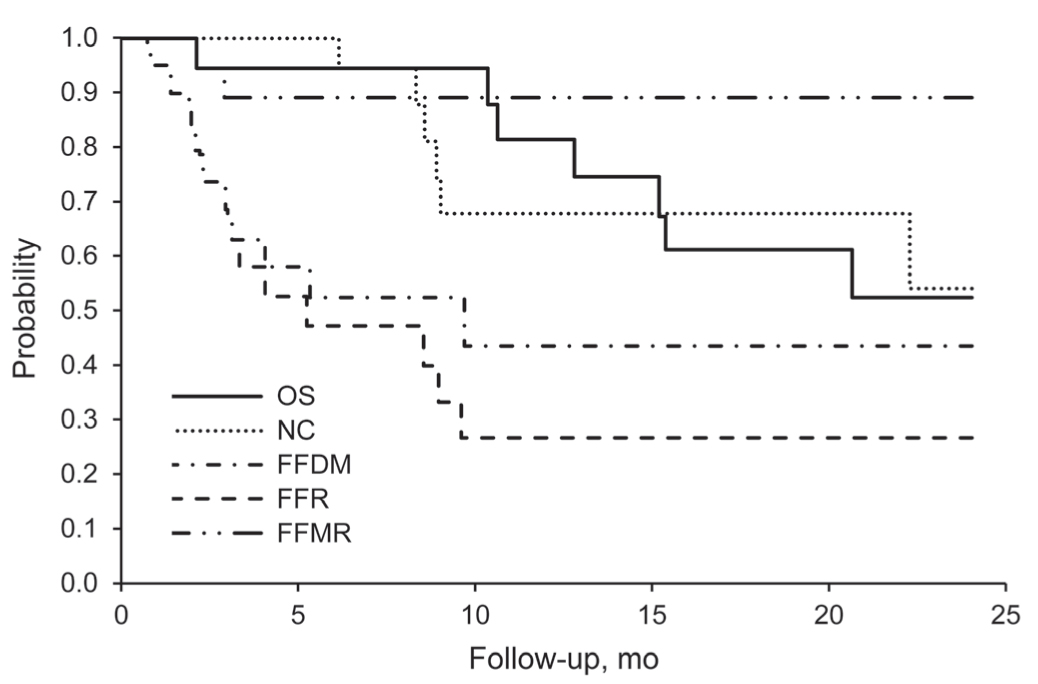
Univariate analysis of several patient and treatment characteristics was performed (Table 4). IMRT was associated with a higher 2-year freedom-from-recurrence rate of 38% compared with 0% for 3-dimensional conformal radiotherapy (P = .01). Factors typically considered prognostic (eg., thickness, node size and number, extracapsular extension) were not significant in our study, likely due to small numbers.
| Characteristic | OS, % | P Value | FFR, % | P Value | NC, % | P Value | FFDM, % | P Value | |
| All patients | 53 | 27 | 54 | 44 | |||||
| Sex | |||||||||
| Male | 46 | 0.72 | 27 | 0.54 | 80 | 0.04a | 49 | 0.22 | |
| Female | 60 | 33 | 21 | 33 | |||||
| Primary tumor site | |||||||||
| Known | 52 | 0.86 | 32 | 0.40 | 44 | 0.95 | 61 | 0.09 | |
| Unknown | 50 | 17 | 67 | 17 | |||||
| Thickness | |||||||||
| ≤4 mm | 80 | 0.1 | 49 | 0.03a | 36 | 0.82 | 65 | 0.44 | |
| >4 mm | 25 | 0 (at 8 mo) | 50 | 50 (at 20 mo) | |||||
| Systemic therapy | |||||||||
| No | 38 | 0.28 | 27 | 0.97 | 67 | 0.77 | 80 | 0.36 | |
| Yes | 56 | 27 | 51 | 37 | |||||
| Node size | |||||||||
| <3 cm | 53 | 0.99 | 25 (at 12 mo) | 0.88 | 100 | 0.14 | 44 (at 12 mo) | 0.57 | |
| 3 cm | 56 | 40 | 43 | 40 | |||||
| Positive nodes | |||||||||
| ≤2 nodes | 63 | 0.13 | 44 | 0.08 | 67 | 0.06 | 55 | 0.24 | |
| >2 nodes | 34 | 0 (at 9 mo) | 35 | 0 (at 9 mo) | |||||
| ECE | |||||||||
| No | 55 | 0.89 | 33 | 0.49 | 33 | 0.15 | 64 | 0.12 | |
| Yes | 51 | 17 (at 12 mo) | 88 | 17 (at 12 mo) | |||||
| RT technique | |||||||||
| 3-D conformal | 40 | 0.58 | 0 (at 8 mo) | 0.01a | 25 | 0.27 | 20 (at 20 mo) | 0.08 | |
| IMRT | 62 | 38 | 74 | 51 | |||||
| RT dose | |||||||||
| <48 Gy | 53 | 0.46 | 25 | 0.99 | 56 | 0.64 | 40 | 0.67 | |
| 48 Gy | 50 (at 21 mo) | 40 (at 19 mo) | 50 (at 19 mo) | 60 (at 19 mo) | |||||
| Timing of S+RT | |||||||||
| Immediate | 64 | 0.20 | 28 | 0.99 | 65 | 0.16 | 35 | 0.33 | |
| Delayed | 30 (at 21 mo) | 25 (at 12 mo) | 33 (at 12 mo) | 67 (at 20 mo) | |||||
| Eligible for TROG trial | |||||||||
| Yes | 62 | 0.23 | 26 | 0.74 | 56 | 0.62 | 37 | 0.45 | |
| No | 0 (at 20 mo) | 33 (at 12 mo) | 50 (at 12 mo) | 67 (at 20 mo) | |||||
Abbreviations: ECE = extracapsular extension; FFDM = freedom from distant metastases; FFR = freedom from recurrence; IMRT = intensity-modulated radiotherapy; NC = nodal control; OS = overall survival; RT = radiotherapy; S = surgery; TROG = Trans-Tasman Radiation Oncology Group; a = Statistically significant.
Recurrence
With a median follow-up of 15 months in surviving patients, 13 patients (65%) had experienced disease progression (Table 5). Distant metastases developed in 8 patients (40%), in-field nodal recurrence in 2 (10%), out-of-field marginal recurrence in 1 (5%), in-field nodal recurrence and distant metastasis in 1 (5%), and out-of-field marginal recurrence and distant metastasis in 1 (5%). In 3 patients who initially experienced distant metastases, in-field nodal recurrences developed eventually, for a total of 6 in-field nodal recurrences. The median time to in-field nodal recurrence for the 6 patients was 8.7 months (range, 2.0-22 months). The first site of distant metastasis was lungs, 4; bone, 6; pancreas, 1; liver, 1; subcutaneous nodules, 2; supraclavicular nodule, 1; and jugular vein, 1.
| Site of disease progression | 3-D conformal (n=5) | IMRT (n=15) | Immediate RT administration (First Line) (n=13) | Delayed RT administration (Recurrence) (n=7) |
| Local | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Distant | 3 (60) | 5 (33) | 7 (54) | 1 (14) |
| Nodal | 1 (20) | 1 (7) | 1 (8) | 1 (14) |
| Nodal+distant | 0 (0) | 1 (7) | 1 (8) | 0 (0) |
| Marginal | 0 (0) | 1 (7) | 0 (0) | 1 (14) |
| Marginal+distant | 1 (20) | 0 (0) | 0 (0) | 1 (14) |
| No disease progression | 0 (0) | 7 (47) | 4 (31) | 3 (43) |
Abbreviations: IMRT = intensity-modulated radiotherapy; RT = radiotherapy; 3-D = 3-dimensional; a = Values are number (percentage).
In 1 of the 2 patients with marginal recurrences, the treatment failure occurred inferior to the target volume, which extended generously beyond the operative bed. In the other patient, the recurrence was inside the operative bed, which was not covered by the target volume.
Treatment-related complications
Treatment-related complications were documented in 14 patients (70%) (Table 6). Nine patients experienced only lymphedema. Of these, 5 patients had grade 1 lymphedema. Two patients had grade 1 lymphedema before the start of radiation without subsequent progression of symptoms. One patient initially had grade 1 lymphedema, which progressed to elbow contracture with a second course of radiotherapy. Another patient had an initial diagnosis of grade 1 lymphedema, which progressed to a “recalcitrant” state. One patient had grade 1 lymphedema before the start of radiation, but fibrosis developed. All patients experiencing lymphedema were treated with a compression garment.
| Complication | Axillary (n=11) | Inguinal (n=9) | 3-D Conformal (n=5) | IMRT (n=15) | <48 Gy (n=15)> | ≥48 Gy (n=5) |
| Lymphedema | 4 (36) | 5 (56) | 2 (40) | 7 (47) | 6 (40) | 3 (60) |
| Lymphedema + fibrosis | 0 (0) | 1 (11) | 0 (0) | 1 (6.67) | 0 (0) | 1 (20) |
| Wound dehiscence | 1 (9) | 0 (0) | 0 (0) | 1 (6.67) | 1 (6.67) | 0 (0) |
| Lymphedema + wound dehiscence | 0 (0) | 1 (11) | 0 (0) | 1 (10) | 1 (6.67) | 0 (0) |
| Lymphedema + seroma | 0 (0) | 1 (11) | 0 (0) | 1 (6.67) | 0 (0) | 1 (20) |
| Lymphedema + seroma + fibrosis | 0 (0) | 1 (11) | 1 (20) | 0 (0) | 1 (6.67) | 0 (0) |
| None | 6 (55) | 0 (0) | 2 (40) | 4 (27) | 6 (40) | 0 (0) |
Abbreviations: IMRT = intensity-modulated radiotherapy; RT = radiotherapy; 3-D = 3-dimensional; a = Values are number (percentage).
One patient had grade 1 lymphedema and an inguinal wound dehiscence. The wound was treated with a free-flap reconstruction (grade 3 complication). One patient had an axillary and shoulder wound dehiscence attributed to radiation recall, initiation of multidrug chemotherapy after radiotherapy, and infection with methicillin-sensitive Staphylococcus aureus. The wound was managed with multiple débridements and 2 free-flap closures (grade 3 complication).
One patient had grade 1 lymphedema and a seroma after initial inguinal lymph node surgery and continued to experience these complications through radiotherapy. This patient had a prior lymph node dissection. After radiotherapy, the lymphedema worsened to the point of “limited mobility,” and the seroma remained unresolved (grade 3 complication). One patient had grade 2 lymphedema documented as “erythema and induration”; however, it was unclear whether the findings were from the lymphedema or from the lymphedema and concurrent inguinal seroma and recurrent cellulitis. This patient had 3 prior inguinal dissections, and fibrosis also developed in this patient.
Interestingly, 3 patients received a prophylactic lymphedema compression stocking but did not have lymphedema documented. There was no brachial or lumbosacral plexopathy. The median time interval between surgery and the start of postoperative radiotherapy did not differ in patients who did and did not have complications: 5.0 weeks (range, 3.3-10.5 weeks) compared with 5.6 weeks (range, 3.0-6.7 weeks respectively).
DiscussionTop
Although melanoma has traditionally been regarded as a radioresistant disease, a contemporary study has shown NC is significantly improved in selected patients treated with adjuvant radiotherapy [5]. We report a 2-year NC rate of 54% for the entire group, with a rate of 54% for patients with axillary metastases and 67% for patients with inguinal metastases. Though our reported time frame for outcomes is different than 5-year published reports, we still see that our NC rate is less than reported NC rates of 81% to 89% for all nodal metastases, 88% to 91% for axillary metastases, and 69% to 77% for inguinal metastases at 5 years (Table 7) [1, 77, 15]. The intergroup randomized Trans-Tasman Radiation Oncology Group (TROG) trial reported that 20 of 109 patients who received adjuvant radiotherapy experienced a recurrence within the lymph node field compared with 34 of 108 observational patients [5]. The 3-year reported cumulative incidence of lymph node relapse was 19% in the radiotherapy group and 31% in the observational group [5]. The higher risk of lymph node field relapse in the observational group was also similar on analysis for axillary and inguinal nodal sites [5]. Therefore, our NC rate of 54% is also less than that in both arms of the TROG trial. The lower NC rate in our study might be a reflection of our cohort’s characteristics. Only 4 of our patients met the eligibility criteria for the TROG study. Three of the patients were treated with adjuvant radiotherapy after 2 or more nodal recurrences. Further, we also reported subsequent nodal recurrences even after distant metastases were documented.
| Study, Year | No. of patients | Era | Median follow-up, mo | LN field | Radiotherapy | No. of relapses | No. of LN relapses/NC rate, % in all patients (Follow-up) | No. of distant relapses/ DMFS, % | No. of deaths/OS, % | Common toxic effects |
| Present study, 2013 | 20 | 2003-2011 | 15 | A, I | Generally 30 Gy/5 fx | 13 | 6/54 (2 y) | 8/44 (2 y) | 10/53 (2 y) | Lymphedema (medically manageable) |
| TROG [5], 2012 | 109 | 2002-2007 | 40 | A, I, H&N | 48 Gy/20 fx | 69 | 20-81 (3 y) | 54/… | 59/… | Seroma, dermatitis |
| Agrawal et al. [1], 2009 | 509 | 1983-2003 | 60 | C, A, I, E | Generally 30 Gy/5 fx | 455 | 52/91 A, 69 I (5 y) | 282/55.4 | 347/44 (5 y) (all patients) | Lymphedema (medically manageable), highest risk for inguinal region |
| Beadle et al. [15], 2008 | 200 | 1984-2005 | 59 | A | Generally 30 Gy/5 fx | 111 | 20/88 (5 y) | …/46 (5 y) | 99/51 (5 y) | Lymphedema |
| Ballo et al. [7], 2005 | 466 | 1983-2003 | 50.3 | C, A, I, E | Generally 30 Gy/5 fx | 252 | 42/89 (87 A, 77 I) (5 y) | 240/44 (5 y) | 221/… | Lymphedema (medically manageable), highest risk for inguinal region |
Abbreviations: A = axillary lymph node; C = cervical lymph node; DMFS = distant metastasis-free survival; E = epitrochlear; fx = fractions; H&N = head and neck; I = inguinal; LN = lymph node; NC = nodal control; OS = overall survival.
With regard to treatment-related complications, there was no plexopathy. Lymphedema was mainly grade 1 and managed by referral to the lymphedema clinic and use of compression garments. Moreover, none of the lymphedema cases were severe enough to cause skin folds. The 2 patients with the poor inguinal, axillary and shoulder wound healing were successfully treated with reconstructive surgery. There was no difference in the time interval between surgery and the start of postoperative radiotherapy between patients who had no complications and those in whom wound dehiscence developed, suggesting there is no relationship between wound-healing issues and the interval between surgery and the onset of radiotherapy in our study.
Nevertheless, the pattern of lymphedema-prominent treatment-related complications in our study is similar to that previously reported, with the greater part of complications being lymphedema and the vast majority of lymphedema being low grade and manageable medically [1, 2, 6, 7, 16]. In the TROG trial, the most common adverse event related to radiotherapy in the axilla and groin was dermatitis, and the most common adverse events related to surgery were seroma formation and wound infection [5]. This relatively small magnitude of severe radiotherapy-induced lymphedema is most likely a reflection of improved radiotherapy and lymphedema management techniques in the modern era. Interestingly, 3 patients in our study received prophylactic compression stockings, and lymphedema did not develop in these patients, suggesting investigation is warranted into whether prophylactic use of compression stockings reduces the incidence and severity of lymphedema.
Although adjuvant radiotherapy has been shown to improve NC in selected patients in a prospective trial, the optimal technique and schedule remain controversial. This study specifically reports outcomes for IMRT. IMRT is more expensive than 3-dimensional conformal radiotherapy and subsequently may be thought of as placing a greater financial burden on the patient and society. However, IMRT allows for more conformal dose to the tumor volume while sparing critical structures such as the brachial plexus in the axilla and the small bowel, bladder, rectum, external genitalia, and hip when treating the inguinal region. However, the higher charge per treatment can be mitigated by the use of hypofractionation with fewer treatments.
With regard to optimal radiotherapy dose and fractionation, there is no single standard. Hypofractionated regimens have been favored by some on the basis of radiobiological data suggesting melanoma may be more responsive to higher-dose-per-fraction regimens, although this has not been confirmed clinically [12]. The TROG trial used 48 Gy in 20 fractions. Retrospective studies have generally analyzed outcomes for hypofractionated regimens and have documented impressive NC rates [2, 77, 9, 16-18]. A hypofractionated regimen is more convenient for patients and also less costly. Future studies could compare 48 Gy in 20 fractions to a more hypofractionated regimen.
Although surgery and adjuvant radiotherapy may satisfactorily control nodal disease, the risk of distant metastases and melanoma-related death was relatively high in the patients reported in this study. We report a 2-year overall survival rate of 53% and freedom from distant relapse rate of 27%. This is less than the 5-year overall survival and freedom from distant relapse rates of 51% and 46% to 49%, respectively, reported by others [7, 16]. As noted above, our higher rates are probably a reflection of selection bias (our referral pattern and/or patient population). The introduction of ipilimumab into the multidisciplinary management of metastatic melanoma has significantly improved survival outcomes [19]. Nevertheless, distant metastasis remains a high risk in patients with advanced disease requiring further improvements in systemic therapy and further investigations into outcomes of combined systemic and local therapies.
ConclusionTop
Our results show outcomes could be improved in patients with melanoma metastatic to axillary or inguinal lymph nodes who receive adjuvant radiotherapy. We recommend evaluating earlier intervention with postoperative adjuvant radiotherapy and hypofractionated treatment protocols. More effective therapeutics that may be safely combined with radiotherapy are also needed. We acknowledge this study has limitations. It was a small cohort with short median follow-up. Additionally, the retrospective nature of this review inherently creates bias and incomplete data. However, this study reports recurrence and treatment-related outcomes specific for hypofractionated IMRT.
Competing interests
The authors declare no conflict of interest.
ReferencesTop
[1] Agrawal S, Kane JM 3rd, Guadagnolo BA, Kraybill WG, Ballo MT (2009) The benefits of adjuvant radiation therapy after therapeutic lymphadenectomy for clinically advanced, high-risk, lymph node-metastatic melanoma. Cancer 115:5836–5844. Article Pubmed
[2] Ballo MT, Bonnen MD, Garden AS, Myers JN, Gershenwald JE, et al. (2003) Adjuvant irradiation for cervical lymph node metastases from melanoma. Cancer 97:1789–1796. Article Pubmed
[3] Bowsher WG, Taylor BA, Hughes LE (1986) Morbidity, mortality and local recurrence following regional node dissection for melanoma. Br J Surg 73:906–908. Article Pubmed
[4] Lee RJ, Gibbs JF, Proulx GM, Kollmorgen DR, Jia C, et al. (2000) Nodal basin recurrence following lymph node dissection for melanoma: implications for adjuvant radiotherapy. Int J Radiat Oncol Biol Phys 46:467–474. Article Pubmed
[5] Burmeister BH, Henderson MA, Ainslie J, Fisher R, Di Iulio J, et al. (2012) Adjuvant radiotherapy versus observation alone for patients at risk of lymph-node field relapse after therapeutic lymphadenectomy for melanoma: a randomised trial. Lancet Oncol 13:589–597. Article Pubmed
[6] Ang KK, Peters LJ, Weber RS, Morrison WH, Frankenthaler RA, et al. (1994) Postoperative radiotherapy for cutaneous melanoma of the head and neck region. Int J Radiat Oncol Biol Phys 30:795–798. Article Pubmed
[7] Ballo MT, Ross MI, Cormier JN, Myers JN, Lee JE, et al. (2006) Combined-modality therapy for patients with regional nodal metastases from melanoma. Int J Radiat Oncol Biol Phys 64:106–113. Article Pubmed
[8] Burmeister BH, Mark Smithers B, Burmeister E, Baumann K, Davis S, et al. (2006) A prospective phase II study of adjuvant postoperative radiation therapy following nodal surgery in malignant melanoma: Trans Tasman Radiation Oncology Group (TROG) Study 96.06. Radiother Oncol 81:136–142. Article Pubmed
[9] Stevens G, Thompson JF, Firth I, O’Brien CJ, McCarthy WH, et al. (2000) Locally advanced melanoma: results of postoperative hypofractionated radiation therapy. Cancer 88:88–94. Article Pubmed
[10] Barranco SC, Romsdahl MM, Humphrey RM (1971) The radiation response of human malignant melanoma cells grown in vitro. Cancer Res 31:830–833. Article Pubmed
[11] Dewey DL (1971) The radiosensitivity of melanoma cells in culture. Br J Radiol 44:816–817. Article Pubmed
[12] Overgaard J (1986) The role of radiotherapy in recurrent and metastatic malignant melanoma: a clinical radiobiological study. Int J Radiat Oncol Biol Phys 12:867–872. Article Pubmed
[13] Stevens G, McKay MJ (2006) Dispelling the myths surrounding radiotherapy for treatment of cutaneous melanoma. Lancet Oncol 7:575–583. Article Pubmed
[14] Sause WT, Cooper JS, Rush S, Ago CT, Cosmatos D, et al. (1991) Fraction size in external beam radiation therapy in the treatment of melanoma. Int J Radiat Oncol Biol Phys 20:429–432. Article Pubmed
[15] Edge SB, Byrd DR, Comptom CC, Fritz AG, Greene FL, et al. (2010) New York (NY): Springer. American Joint Committee on Cancer: AJCC cancer staging manual, 7th edition. Article
[16] Beadle BM, Guadagnolo BA, Ballo MT, Lee JE, Gershenwald JE, et al. (2009) Radiation therapy field extent for adjuvant treatment of axillary metastases from malignant melanoma. Int J Radiat Oncol Biol Phys 73:1376–1382. Article Pubmed
[17] Ballo MT, Strom EA, Zagars GK, Bedikian AY, Prieto VG, et al. (2002) Adjuvant irradiation for axillary metastases from malignant melanoma. Int J Radiat Oncol Biol Phys 52:964–972. Article Pubmed
[18] Chang DT, Amdur RJ, Morris CG, Mendenhall WM (2006) Adjuvant radiotherapy for cutaneous melanoma: comparing hypofractionation to conventional fractionation. Int J Radiat Oncol Biol Phys 66:1051–1055. Article Pubmed
[19] Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, et al. (2010) Improved survival with Ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723. Article Pubmed


