Journal of Cancer Research & Therapy
An International Peer-Reviewed Open Access Journal
ISSN 2052-4994


- Download PDF
- |
- Download Citation
- |
- Email a Colleague
- |
- Share:
-
- Tweet
-

Journal of Cancer Research & Therapy
Volume 2, Issue 7, July 2014, Pages 105-110
Case reportOpen Access
Pseudocystic pheochromocytoma: An incidental finding and a rare entity
-
Petramala L1,
Zinnamosca L1,
Marinelli C1,
Calvieri C1,
Settevendemmie A1,
Concistrè A1,
Salvi D1,
Ciardi A2,
D’Ermo G2,
De Toma G2 and
Letizia C1,*

- 1 Specialized Center of Secondary Hypertension, Department of Internal Medicine and Medical Specialties, University of Rome “LA Sapienza, Rome, Italy
- 2 Department of Surgery “Pietro Valdoni”, University of Rome “LA Sapienza, Rome, Italy
*Corresponding author: Prof. Claudio Letizia, M.D., Department of Internal Medicine and Medical Specialties, Policlinico Umberto I, Viale del Policlinico 155, 00165, Rome, Italy. Email: claudio.letizia@uniroma1.it
Received 19 April 2014 Revised 15 June 2014 Accepted 22 June 2014 Published 29 June 2014
DOI: http://dx.doi.org/10.14312/2052-4994.2014-16
Copyright: © 2014 Petramala L, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
AbstractTop
Cystic adrenal neoplasms are uncommon and may be incidentally discovered during radiologic procedure not directly performed for the evaluation of adrenal disease. Cystic pheochromocytomas are uncommon neoplasms that originate from the adrenal medulla, clinically differing from the more frequent solid pheochromocytomas. Here, we report a case of 72 year-old woman affected by asymptomatic pseudocystic pheochromocytoma (70x50mm), incidentally discovered, with a false negative 123I-monoiodobenzylguanidine (123I-MIBG) scintigraphy. Histopathological examination led to diagnosis of pseudocystic pheochromocytoma with typical architecture. The screening for germline mutation of succinate dehydrogenase complex subunits (SDHB, SDHC), Von Hipple-Lindau syndrome (VHL) and multiple endocrine neoplasia type 2 (RET) genes were negative, finding only some polymorphisms for RET and SDHB. We emphasize the relevance of an accurate pre-operative diagnosis, especially in rare diseases, and the requirement of experience and multidisciplinary management in these rare pathologies.
Keywords: adrenal cyst; pseudocystic pheochromocytoma; adrenal incidentaloma; false-negative 123I-monoiodobenzylguanidine (MIBG) scintigraphy
IntroductionTop
Cystic adrenal neoplasms are uncommon and may be discovered incidentally during a radiologic procedure not performed for the evaluation of adrenal disease. Adrenal pseudocysts accounts for about 40% of adrenal cysts [1], and three mechanisms for their formation are hypothetical: a) cystic degeneration of primary adrenal neoplasm; b) degeneration of a vascular neoplasm, and c) malformation and hemorrhage of adrenal veins into the adrenal gland [2].
Cystic pheochromocytomas are uncommon neoplasms that originate from the adrenal medulla, and differing from the more frequent solid pheochromocytomas, which present episodic headache, diaphoresis, palpitations and hypertension, and may be clinically silent [3, 4].
Here, we report a case of asymptomatic pseudocystic pheochromocytoma, incidentally discovered, with a false negative 123I-monoiodobenzylguanidine (123I-MIBG) scintigraphy.
Case reportTop
A 72 year-old woman was admitted to our center with vague pain in abdomen and a well-defined mass (56x31 mm) localized between the right liver lobe and the right kidney at ultrasonography, attributable to the right adrenal gland.
The medical history reported type 2 diabetes mellitus, obesity, post-thyroiditis hypothyroidism and hypercholesterolemia. The patient was taking metformin, statin and levothyroxine. At clinical examination she did not show any sign of abdominal tumefaction or tenderness. The physical findings were characterized by 160 cm tall, weighed 79 kg, waist circumference (WC) 107 cm, and body mass index (BMI) 30.5 kg/m2. Clinical blood pressure was 135/85 mmHg in clinostatism and in orthostatism, and heart rate was normal (68 beats/min) with sinus rhythm. Chest X- ray did not demonstrate pulmonary congestion and arterial blood gases were as follows: pH 7.40; PO2 91; PCO2 20; HCO3- 18.1. Laboratory investigations including blood routine, electrolytes and chemistry were reported in Table 1. Ambulatory blood pressure monitoring (ABPM) revealed diurnal and nocturnal normal blood pressure values with dipper profile. Plasma renin activity (PRA), plasma aldosterone concentration (PAC), and PAC/PRA ratio were normal (Table 2). Normal levels of plasma cortisol (PC), plasma ACTH, 24 h urinary free cortisol (UFC) and aldosterone excretions were found. Increased level of plasma chromogranin A (CgA), 24 h urinary free metanephrines and vanillymandelic acid (VMA) excretions were revealed (Table 2).
| Laboratory test | Results | Normal values |
| Haematology | ||
| White blood cells | 4.71 | 4.40-11.30/mm3 |
| Neutrophils | 45.6 | 40.90-68.10 % |
| Eosinophils | 3.9 | 0.00-5.40 % |
| Basophils | 1.5 | 0.00-1.20 % |
| Monocytes | 6.2 | 3.50-10.50 % |
| Lymphocytes | 39.6 | 24.90-40.10 % |
| Red blood cells | 4.48 | 4.30-5.90/µL |
| Haemoglobin | 13.2 | 13.5-16.50 g/dL |
| Haematocrit | 40.3 | 40-52 % |
| Platelets | 272 | 150-450/µL |
| Biochemistry | ||
| Creatinine | 0.7 | 0.7-1.2 mg/dL |
| Sodium | 140 | 136-145 mmol/L |
| Potassium | 4.3 | 3.4-5.5 mmol/L |
| Chloride | 104 | 98-109 mmol/L |
| Calcium | 9.88 | 8.4-10 mg/dL |
| Blood urea nitrogen | 39 | 10.2-49.8 mg/dL |
| Total proteins | 70 | 60-82 g/L |
| Albumins | 47 | 35-55 g/L |
| Aspartate aminotransferase | 19 | 9-45 U/L |
| Alanine aminotransferase | 6 | 10-40 U/L |
| Lactate dehydrogenase | 141 | 135-225 U/L |
| Amylase | 72 | 28-100 U/L |
| Blood fasting glucose | 102.7 | 70.3-100.9 mg/dL |
| Laboratory findings | Pre-operative results | Post-operative results | Normal values |
| Plasma serum | |||
| Plasma renin activity (PRA) | 0.1 | 0.7 | 0.2-2.7 ng/ml/h |
| Plasma aldosterone (PAC) | 58.7 | 73.8 | 30-160 pg/ml |
| Serum cortisol (PC) | 8.9 | 9.67 | 9.64-26.09 µg/dL |
| Plasma ACTH | 30 | 29 | 10-90 pg/ml |
| Serum DEAS | 90.9 | 123.4 | 61.2-511.7 µg/dl |
| Serum 17 α-OH-progesterone | 1.7 | 1.7 | 0.08-1.8 ng/mL |
| Serum chromogranine A (Cg A) | 108 | 65 | <90 g/mL |
| Serum cortisol after DXM (1 mg) | 0.7 | 0.7 | <1.8 µg/dL |
| Urine | |||
| Aldosterone | 8.7 | 5.3 | 2.84-34 µg/24h |
| Cortisol | 83.7 | 112.2 | 38-208 µg/24h |
| Metanephrines | 1600 | 4.7 | 20-345 µg/24h |
| Vanillylmandelic acid | 15 | 9.5 | <13.6 mg/24h |
The patient with suspected of adrenal pheochromocytoma, underwent a computed tomography (CT) scan of abdomen and a retrocaval right adrenal mass (60x50 mm in diameter) with regular walls, thin septa and increased contrast enhancement was showed (Figure 1). There were no other pathological findings in the abdomen. Moreover, an 123I-MIBG scintigraphy revealed no abdominal pathological accumulation (Figure 2). However, these findings meant that it was still possible that the right adrenal mass was a pheochromocytoma, which could have been dormant at that time but could, upon being triggered due to release of catecholamine. However, the adrenal mass was larger than 60 mm in diameter, and elective surgery was planned based on a multidisciplinary approach. We decided on open surgery to remove the right adrenal mass. Our surgeons performed exploratory laparotomy via midline under general anesthesia in elective conditions with appropriate therapy (alpha adrenergic receptor blockade and plasma expansion) with a good control of blood pressure. Exploration of abdomen revealed a 70 x 50 mm dark lesion originates from the right adrenal gland and was completely excised with adrenalectomy (Figure 3). No cardiovascular alterations were found during operation.
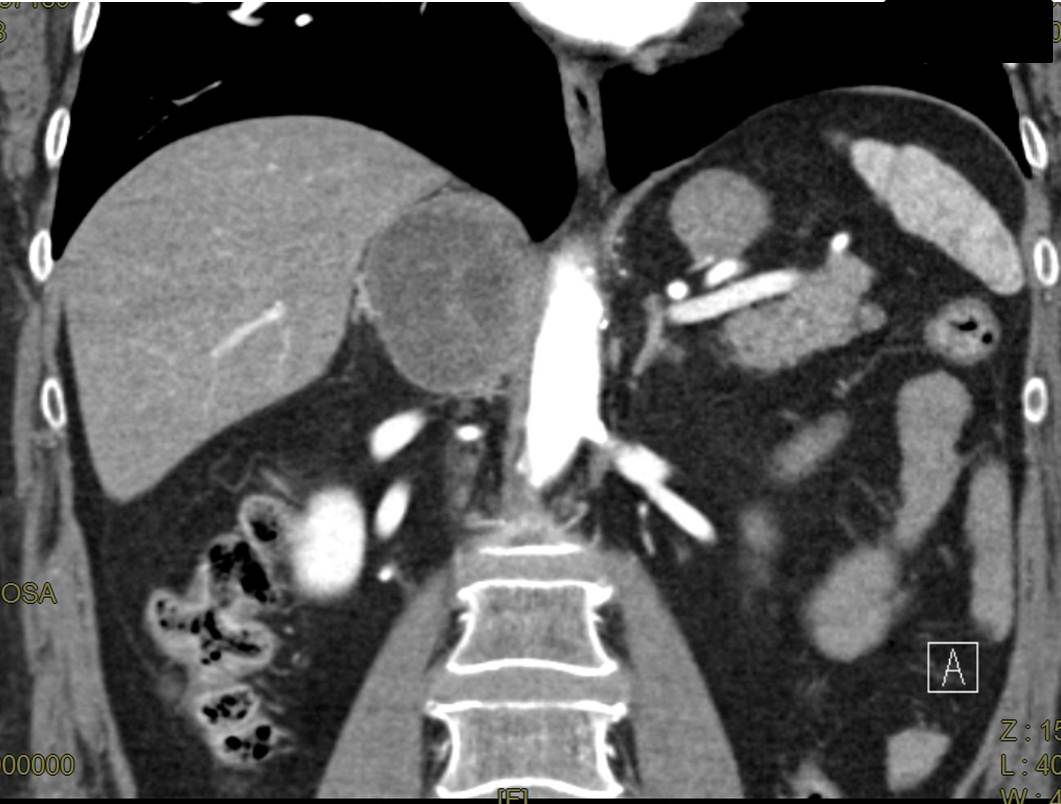
Figure 1 Abdominal computed tomography (CT): retrocaval right adrenal mass (60x50 mm in diameter) with regular walls, thin septa and increased contrast enhancement.

Figure 2 123I-monoiodobenzylguanidine (123I-MIBG) scintigraphy: Absence of abdominal pathological accumulation..
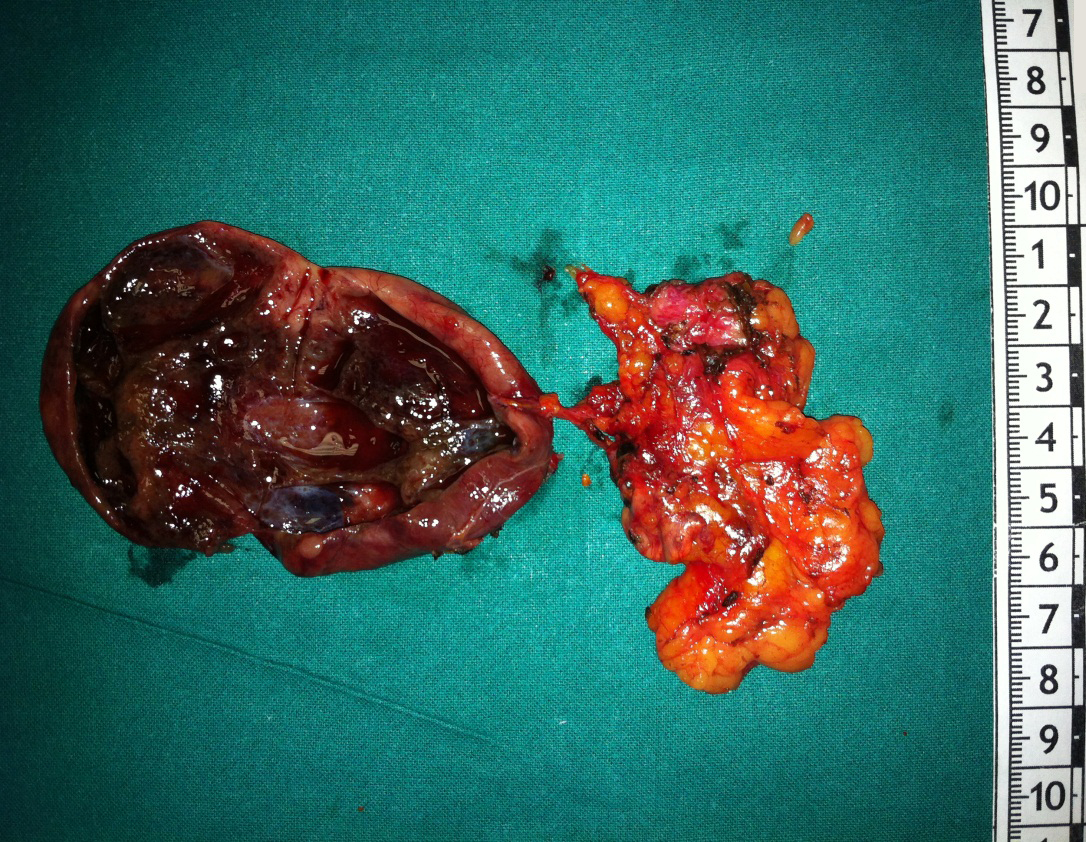
Figure 3 Abdominal surgical exploration: dark lesion from the right adrenal gland (70 x 50 mm).
Histopathological examination of the adrenal lesion revealed a mass, measuring 65 x 50 x 50 mm in diameter with wall of 0.7 cm, without necrotic areas. Histological picture shows the wall of giant pseudocystic pheochromocytoma with typical architecture of “zellballen” nests, and peripheral sustentacular cells (Figure 4 A, B). A diagnosis of an adrenal pseudocystic pheochromocytoma was made. The patient’s post-operative course was uneventful and she was discharged ten days after the operation. Follow-up demonstrated that the patient remains well, ABPM was normal, and was asymptomatic five months after surgery with normal laboratory data (Table 2).
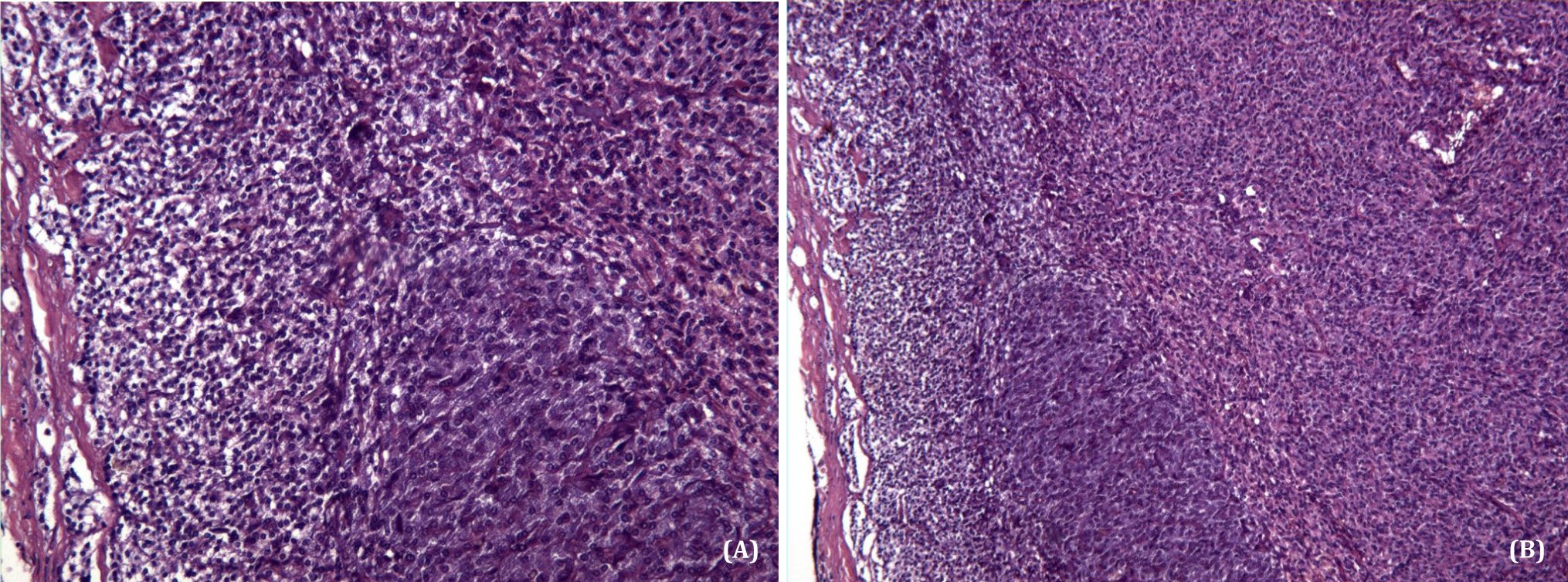
Figure 4A, B Histyological picture shows the wall of pseudocystic pheochromocytoma with typical architecture of “zellballen” nests, with peripheral sustentacular cells (H&E, A 100x; B 250x).
Patient’s informed consent for the publication of patient’s data and images was obtained, subsequently all the major susceptibility genes were assayed with genetic testing. Screening for germline mutation of SDHB, SDHC and VHL genes was negative. We therefore decided to perform additional tests such as RET gene. We only revealed some polymorphisms of SDHB and RET genes (Table 3).
| Gene RET | es 11 | cod 691 | GGT>AGT | Gly>Ser eter |
| Gene RET | es 13 | cod 769 | CTT>CTG | Leu>Leu eter |
| Gene RET | es 14 | cod 836 | AGC>AGT | Ser>Ser eter |
| Gene RET | es 15 | cod 904 | TCC>TCG | Ser>Ser eter |
| Gene SDHB | es 5 | cod 163 | TCT>CCT | Ser>Pro eter |
DiscussionTop
Adrenal tumors represent one of the most fascinating fields of contemporary endocrine research. Due to the advances in imaging techniques, the incidence of incidentally discovered adrenal masses (incidentalomas), is strictly increasing [5, 6]. Most of these incidentalomas are benign, hormonally inactive tumors of adrenocortical origin. Hormone secreting adrenocortical adenomas may lead to severe diseases with significant morbidity and mortality including hypercortisolism or primary aldosteronism [7, 8]. Despite being a rare tumor, adrenocortical cancer is a clinically relevant disease, since its prognosis is very poor in advanced stages [9].
Tumors arising from adrenal medullary tumors, as pheochromocytomas, are responsible for an important and potentially curable cause of secondary hypertension [10-13]. They are solid and well vascularized neoplasm, and very rarely they may present as predominantly cystic mass [14]. Many patients with catecholamines secreting tumors do not show the classical symptoms such as headache, palpitations and diaphoresis, and approximately half have normal blood pressure [15-17]. Moreover, their symptoms are generally associated with an abdominal mass or even pain, particularly if the mass attains large dimensions. In this report we described a patient with incidentally discovered giant pseudocystic pheochromocytoma that no displayed the classical symptoms of the adrenergic syndrome, but her initial presentation was an abdominal pain with discomfort.
When an adrenal mass is incidentally detected, the occurrence of any subclinical cortisol or aldosterone secreting lesion, and a pheochromocytoma is excluded. Moreover, to resect all adrenal non-functioning lesions, which are suspicious for adrenocortical carcinoma at imaging, because fine needle aspiration biopsy (FNAB) does not always allow to distinguish from malignant primary adrenocortical cancers [18, 19]. Adrenal cysts are infrequently observed, and some cases have been reported in the literature [20-22]. In particular, the first report describes a case with bilateral pheochromocytoma presenting as primarily cystic suprarenal masses [20]. The second case is a large pseudocystic pheochromocytoma associated with pneumatosis cystoides intestinalis [21]. The third case is a 64 year-old man with right flank pain, hematuria and pseudocystic adrenal pheochromocytoma [22].
Cysts of adrenal gland are usually non-functional, asymptomatic and incidental findings, and are histologically clarified in endothelial cyst, pseudocyst and infectious cyst [20]. Adrenal pseudocyst consists of a fibrous wall without a cellular lining [23], and their etiology remains today uncertain. An hypothesis include hemorrhage into adrenal gland parenchyma secondary to trauma, cystic degeneration of primary adrenal neoplasm and a vascular neoplasm or malformation [24-25].
In a total of 41 cases of macroscopically cystic lesions among patients who underwent at the Mayo Clinic, experienced over a 25 years period, Erickson and coworkers [26] identified 32 adrenal pseudocysts, 8 endothelial cysts and 1 epithelial cyst. In particular, of the 32 pseudocysts, 6 were associated with adrenal neoplasm, including 2 adrenocortical carcinomas, 2 adrenal adenomas and 2 pheochromocytomas. Moreover, Sebastiano C at al. [27] in their 20 years’ experience (1992-2012) identified 30 cases of adrenal lesions exhibiting a significant cystic component. In this case series, 12 cases were pseudocysts without any inner wall lining or associated neoplastic tissue, 2 were endothelial derived, and 17 were epithelium/neuroectoderm-derived cystic neoplasm. Of these 17 epithelium/neuroectoderm-derived cystic neoplasm, 16 were benign pheochromocytomas, 2 of which were bilateral lesions in the same patient. All of these 16 cases of pheochromocytoma exhibited adjacent compressed benign adrenal tissue. In this series, cystic and pseudocystic non symptomatic pheochromocytoma are rarely reported [27].
Recently, Kyoda et al. [28], in 31 patients with radiographically diagnosed pheochromocytoma, experienced three cases of adrenal pseudocysts that were pre-operatively diagnosed as pheochromocytoma. In particular MIBG scintigraphy was negative in all of the pseudocystic patients, but positive in all adrenal solid pheochromocytoma patients. In our case 123I-MIBG scintigraphy did not show abnormal findings and did not provide additional information for diagnosis of adrenal pheochromocytoma.
Adrenal medullary scintigraphy employs the use of radioiodine guanethidine derivate such as 131I-MIBG and 123I-MIBG. Both tracers combined have shown to have increased specificity (95%), but the sensitivity is only 80% to 90% [29-31]. However, tumors less than 1.5 cm in diameter, and with extensive necrosis or hemorrhage may yield false negative results due to poor uptake of the tracer with the possibility of false negative results scintigraphy should be complemented with hormonal evaluation and imaging studies to establish accurate diagnosis.
MIBG can also point to additional lesions not indicated by conventional imaging in patients with metastatic disease or multifocal disease associated with mutations of disease-susceptibility genes [32]. Moreover, a false negative 123I-MIBG is frequently related to metastatic tumors and usually due to SDHB mutations with unfavourable prognosis [33].
In our patient we performed genetic tests (SDHD, SDHB, VHL and RET) [34] but we revealed non-genetic mutations. Finally, the guidelines of the American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons do not recommend routine use of MIBG scintigraphy [35].
ConclusionTop
The pseudocystic and asymptomatic adrenal pheochromocytoma is rare. We would like to emphasize importance of pre-operative diagnosis, and the treatment in these forms requires experience and multidisciplinary management.
Conflict of interest
The authors wish to express that they have no conflict of interest.
ReferencesTop
[1] Amarillo HA, Bruzoni M, Loto M, Castagneto GH, Mihura ME (2004) Hemorrhagic adrenal pseudocyst: laparoscopic treatment. Surg Endosc 18:1539. Article Pubmed
[2] Habra MA, Feig BW, Waguespack SG (2005) Image in endocrinology: adrenal pseudocyst. J Clin Endocrinol Metab 90:3067-3068. Article Pubmed
[3] Rao A, Majmudar B, Bumpers H (2007) Giant adrenal pseudocyst. Surg Roundas 30:210.
[4] White R, Sinnenberg R (1984) Adrenal pseudocyst in a hypertensive patient. South Med J 77:1189-1191. Article Pubmed
[5] Suga H, Inagaki A, Ota K, Taguchi S, Kato T, et al. (2003) Adrenal pseudocyst mimicking a pheochromocytoma found after a traffic accident. Intern Med 42:66-71. Article Pubmed
[6] Grumbach MM, Biller BM, Braunstein GD, Campbell KK, Carney JA, et al. (2003) Management of the clinically inapparent adrenal mass ("incidentaloma"). Ann Intern Med 138:424-429. Article Pubmed
[7] Angeli A, Osella G, Alì A, Terzolo M (1997) Adrenal incidentaloma: an overview of clinical and epidemiological data from the National Italian Study Group. Horm Res 47:279-283. Article Pubmed
[8] Aron D, Terzolo M, Cawood TJ (2012) Adrenal incidentalomas. Best Pract Res Clin Endocrinol Metab 26:69-82. Article Pubmed
[9] Young WF Jr (2007) Clinical practice. The incidentally discovered adrenal mass. N Engl J Med 356:601-610. Article Pubmed
[10] Mansmann G, Lau J, Balk E, Rothberg M, Miyachi Y, et al. (2004) The clinically inapparent adrenal mass: update in diagnosis and management. Endocr Rev 25:309-340. Article Pubmed
[11] Därr R, Lenders JW, Hofbauer LC, Naumann B, Bornstein SR, et al. (2012) Pheochromocytoma - update on disease management. Ther Adv Endocrinol Metab 3:11-26. Article Pubmed
[12] Manger WM (2009) The protean manifestations of pheochromocytoma. Horm Metab Res 41:658-663. Article Pubmed
[13] Mantero F, Terzolo M, Arnaldi G, Osella G, Masini AM, et al. (2000) A survey on adrenal incidentaloma in Italy. Study Group on Adrenal Tumors of the Italian Society of Endocrinology. J Clin Endocrinol Metab 85:637-644. Article Pubmed
[14] Pradeep PV, Mishra AK, Aggarwal V, Bhargav PR, Gupta SK, et al. (2006) Adrenal cysts: an institutional experience. World J Surg 30:1817-1820. Article Pubmed
[15] Antedomenico E, Wascher RA (2005) A case of mistaken identity: giant cystic pheochromocytoma. Curr Surg 62:193-198. Article Pubmed
[16] Cotesta D, Petramala L, Serra V, Pergolini M, Crescenzi E, et al. (2009) Clinical experience with pheochromocytoma in a single centre over 16 years. High Blood Press Cardiovasc Prev 16:183-193. Article Pubmed
[17] Manger WM (2005) The vagaries of pheochromocytomas. Am J Hypertens 18:1266-1270. Article Pubmed
[18] Pacak K, Eisenhofer G, Ahlman H, Bornstein SR, Gimenez-Roqueplo AP, et al. (2007) Pheochromocytoma: recommendations for clinical practice from the First International Symposium. October 2005. Nat Clin Pract Endocrinol Metab 3:92-102. Article Pubmed
[19] Mazzaglia PJ, Monchik JM (2009) Limited value of adrenal biopsy in the evaluation of adrenal neoplasm: a decade of experience. Arch Surg 144:465-470. Article Pubmed
[20] Falappa P, Mirk P, Rossi M, Troncone L, Butti A, et al. (1980) Bilateral pseudocystic pheochromocytoma. J Comput Assist Tomogr 4:860-862. Article Pubmed
[21] Alanen A, Kormano M (1982) Pseudocystic pheochromocytoma associated with pneumatosis cystoides intestinalis. Case report. Eur J Radiol 2:50-51. Pubmed
[22] G Kojima Y, Sugao H, Yokokawa K, Takiuchi H, Sakurai T, et al. (1988) A case of cystic pheochromocytoma. Hinyokika Kiyo 34:1201-1205. Pubmed
[23] Fukushima N, Oonishi T, Yamaguchi K, Fukayama M (1995) Mesothelial cyst of the adrenal gland. Pathol Int 45:156-159. Article Pubmed
[24] Chew SP, Sim R, Teoh TA, Low CH (1999) Haemorrhage into non-functioning adrenal cysts--report of two cases and review of the literature. Ann Acad Med Singapore 28:863-866. Article Pubmed
[25] Groben PA, Roberson JB Jr, Anger SR, Askin FB, Price WG, et al. (1986) Immunohistochemical evidence for the vascular origin of primary adrenal pseudocysts. Arch Pathol Lab Med 110:121-123. Pubmed
[26] Erickson LA, Lloyd RV, Hartman R, Thompson G (2004) Cystic adrenal neoplasms. Cancer 101:1537-1544. Article Pubmed
[27] Sebastiano C, Zhao X, Deng FM, Das K (2013) Cystic lesions of the adrenal gland: our experience over the last 20 years. Hum Pathol 44:1797-1803. Article Pubmed
[28] Kyoda Y, Tanaka T, Maeda T, Masumori N, Tsukamoto T (2013) Adrenal hemorrhagic pseudocyst as the differential diagnosis of pheochromocytoma-a review of the clinical features in cases with radiographically diagnosed pheochromocytoma. J Endocrinol Invest 36:707-711. Article Pubmed
[29] Shapiro B, Copp JE, Sisson JC, Eyre PL, Wallis, et al. (1985) Iodine-131 metaiodobenzylguanidine for the locating of suspected pheochromocytoma: experience in 400 cases. J Nucl Med 26:576-585. Article Pubmed
[30] Roelants V, Goulios C, Beckers C, Jamar F (1998) Iodine-131-MIBG scintigraphy in adults: interpretation revisited? J Nucl Med 39:1007-1012. Article Pubmed
[31] Miskulin J, Shulkin BL, Doherty GM, Sisson JC, Burney RE, et al. (2003) Is preoperative iodine 123 meta-iodobenzylguanidine scintigraphy routinely necessary before initial adrenalectomy for pheochromocytoma? Surgery 134:918-922. Article Pubmed
[32] Bhatia KS, Ismail MM, Sahdev A, Rockall AG, Hogarth K, et al. (2008) 123I-metaiodobenzylguanidine (MIBG) scintigraphy for the detection of adrenal and extra-adrenal phaeochromocytomas: CT and MRI correlation. Clin Endocrinol (Oxf) 69:181-188. Article Pubmed
[33] Fonte JS, Robles JF, Chen CC, Reynolds J, Whatley M, et al. (2012) False-negative ¹²³I-MIBG SPECT is most commonly found in SDHB-related pheochromocytoma or paraganglioma with high frequency to develop metastatic disease. Endocr Relat Cancer 19:83-93. Article Pubmed
[34] Mannelli M, Castellano M, Schiavi F, Filetti S, Giacchè M, et al. (2009) Clinically guided genetic screening in a large cohort of italian patients with pheochromocytomas and/or functional or nonfunctional paragangliomas. J Clin Endocrinol Metab 94:1541-1547. Article Pubmed
[35] Zeiger MA, Thompson GB, Duh QY, Hamrahian AH, Angelos P, et al. (2009) The American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons medical guidelines for the management of adrenal incidentalomas. Endocr Pract 1:1-20. Article Pubmed


